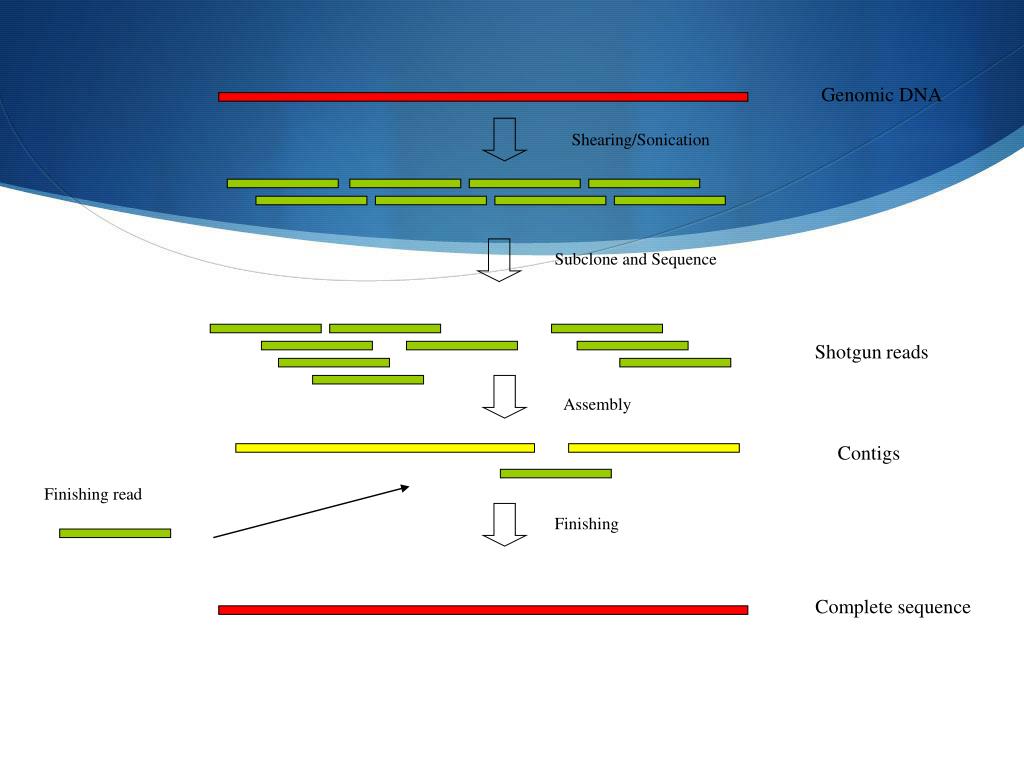
Verify you have trimmed your reads to the best they can be using fastq, multiqc, and trimmomatic.If you don't think the contigs you have are "good enough".If you built your contigs based on a normal/control sample you can map other reads to the contigs using bowtie2 to try to identify variants in other samples.You can turn the contigs.fa into a blast database ( formatdb or makeblastdb depending on which version of blast you have) or try multiple sequence alignments through NCBIs blast.Think about what question you are trying to answer. Look for things: If you're just after a few homologs, an operon, etc.What comes next when working with your own data? Since each is >3000 bases, contigs cannot be connected across them using this data. There are 7 nearly identical ribosomal RNA operons in E. There are additional scripts that change many of the default options such as metaspades.py, plasmidspades.py, and rnaspades.py or these options can be set from the main spades.py script with the flags -meta, -plasmid, -rna respectively. SPAdes is actually written in python and the base script name is "spades.py". As always its a good idea to get a look at what kind of options the program accepts using the -h option. Now let's use SPAdes to assemble the reads. More often (and everywhere else in this course) your read pairs will be "separate" with the corresponding paired reads at the same index in two different files (each with exactly the same number of reads). Dataĭownload the paired end fastq files which have had their adapters trimmed from the $BI/gva_course/Assembly/ how the pairs of reads are denoted by the /1 and /2 at the end of the first line in the 4 line fastq block. I suggest analyzing this data on an idev node and then submitting the other data analysis for the bacterial genomes as a job to run overnight. Fortunately for this class, we can make use of the plasmid spades option to assemble and even smaller plasmid genome that is ~2000 bp long in only a few minutes. If you still run into memory problems, consider moving onto the 'large-mem' queue rather than the 'normal' queue which has more memory, and also downsampling your data.Īssembling even small bacterial genomes can be incredibly time intensive (as well as memory intensive as highlighted above). Unlike other times in the class where we are concerned about being good TACC citizens and not hurting other people by the programs we run, assembly programs are exceptionally memory intensive and attempting to run on the head node may result in the program returning a memory error rather than useable results. When it comes time to assemble your own reference genome, remember to give each sample its own compute node rather than having multiple samples split a single node. 
Other potential tools to have in the same environment would be read preprocessing tools, in particular adapter removal tools such as trimmomatic. Find proteins of interest in an assembly using Blast.Īs genome assembly is important part of analysis but is building a reference file that will be used many times, it makes more sense to install it its own environment.Use contig_ to display assembly statistics.Run SPAdes to perform de novo assembly on fragment, paired-end, and mate-paired data.reads lacking adapters in this case.įor those looking for a real challenge, go through the multiqc tutorial and the trimmomatic tutorial, and use the information provided here to compare assemblies of some of the same samples in both cases. If using this tutorial on your own samples make sure you are working with the best data possible. While not explicitly covered here, the presence of adapter sequences on reads when trying to assemble them can significantly complicate assembly and harm it.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
